Cash in on medical breakthroughs
Genetics is continuing to revolutionise medicine, and is throwing up some exciting investment opportunities. Eoin Gleeson examines the sector, and picks the best way to cash in.
Get the latest financial news, insights and expert analysis from our award-winning MoneyWeek team, to help you understand what really matters when it comes to your finances.
You are now subscribed
Your newsletter sign-up was successful
Want to add more newsletters?

Twice daily
MoneyWeek
Get the latest financial news, insights and expert analysis from our award-winning MoneyWeek team, to help you understand what really matters when it comes to your finances.

Four times a week
Look After My Bills
Sign up to our free money-saving newsletter, filled with the latest news and expert advice to help you find the best tips and deals for managing your bills. Start saving today!
Genetics is continuing to revolutionise medicine, and is throwing up some exciting investment opportunities, says Eoin Gleeson.
America's poor are spitting blood. For the past month they've been herding into town halls to howl their disapproval at Barack Obama's plans to reform healthcare. What are they so angry about? To many, particularly outside the US, it looks as though Obama's plan to extend medical insurance to 47 million uninsured poor Americans has their best interests at heart. So is this a case of the turkeys voting for Christmas? Not at all. Most of the Americans who fought for seats at these healthcare debates are fiercely proud of their healthcare system. They don't want it to change. And that's not because they are a band of blind jingoists, paralysed by fear of Communists in Washington.
Consider this. In 2006, the US accounted for 82% of world research spending in biotechnology, according to the British Medical Journal. A recent report by technology consultants Cels places the top five institutes for stem cell research in America. Between them, the top five US hospitals carry out more clinical trials each year than all the hospitals in any other country combined. It may be hugely wasteful in many ways, yet no other country contributes more to pushing back the frontiers of medicine than America.
MoneyWeek
Subscribe to MoneyWeek today and get your first six magazine issues absolutely FREE

Sign up to Money Morning
Don't miss the latest investment and personal finances news, market analysis, plus money-saving tips with our free twice-daily newsletter
Don't miss the latest investment and personal finances news, market analysis, plus money-saving tips with our free twice-daily newsletter
And that's important. We are living in an era of remarkable medical breakthroughs, but breakthroughs are exactly what we need to combat the diseases that will kill more of us as life expectancy increases. The easy cures treatments have been picked off by the pharmaceutical industry over the past 60 years. The good news is that having decoded the human genome which stores our biological programme across 23 pairs of chromosomes we are starting to learn the secrets of even the most complex diseases. Stem cell labs from Newcastle to Jerusalem break news of stunning new developments by the week. Today's medical industry is one where machines can sequence our genomes, blood from an umbilical cord is used to regenerate the brain, and robots perform remarkable feats of surgery.
In America alone, some $39bn was allocated to healthcare spending in the stimulus bill. Much of that will go to 15,000 additional grants submitted by scientists at universities across the US. Here's how you can benefit from this and from the global surge in medical research.
Decoding our genes
A cousin of mine was born with cystic fibrosis (CF) a hereditary condition that exposes the sufferer to chronic respiratory infections due to an imbalance of salt and water in the lungs. In the early 1980s, doctors struggled to control the effects of CF. My cousin was exposed to a blunderbuss of respiratory drugs and was never far from a mechanical respirator. At that time the average life expectancy for someone with CF was 29.
Today, CF sufferers have a much better chance of making it past their 29th birthday. Scientists have been able to pinpoint the gene that causes CF and are even attempting to replace the faulty versions in patients. When they are successful so far patients' immune defences have blocked treatments it will add years to the lives of young CF sufferers.
Ever since upstart biologist Craig Venter stunned the science community by sequencing a human genome seven years ago, the prospect of a leap in our understanding of human disease has seemed tantalisingly close. By deconstructing the human body cell by cell, scientists believe they will uncover the genetic roots of complex diseases. Many thought the Human Genome Project, sequencing a single genome at the cost of $4bn, would herald cures for everything from cancer to diabetes. What they have actually learned is that these diseases are far more complex than they had imagined.
But scientists are close to a remarkable medical milestone. Dr Venter's big achievement was to sequence a single genome under his own steam for a fraction of the cost of the official Human Genome Project his project cost $100m. Yet now radical improvements in sequencing technology allow scientists to do the job for just $50,000. "I spent ten years searching for just one gene," Dr Venter told The Economist. "Today anyone can do it in 15 seconds."
Scientists have a single target in mind: $1,000 to sequence a genome. "At that price, we enter the era of consumer genetics," says Professor Steven Pinker in The New York Times. We will pay to learn how likely we are to develop hereditary diseases, such as Alzheimer's or Parkinson's, and adjust our lifestyles accordingly. This shift towards preventative medicine heralds a radical evolution for healthcare. A recent study by the American Cancer Society concludes that prevention and early detection decreased cancer death rates between 1990 and 2005 by 19% for men and 11% for women.
At $1,000 to sequence a genome, it will also be cheap enough for widespread medical use. Newborns could be sequenced at birth for medical records; cancer patients will have tumours sequenced as they are developing. That then raises the prospect of personalised medicine, says JP Morgan analyst Tycho Peterson bringing the right drug to the right patient with the right dose, instead of the horrible trial and error that CF patients, for example, have had to cope with in the past. That's why Obama has included $10.4bn to invest in new life-science research over the next two years in his stimulus plans.
But let's not get carried away. It could be up to a decade before the $1,000 target is reached, reckons The Economist. Until then, the primary use of gene data will be in discovering new drugs.
Biologics are drugs made from living sources, such as antibodies, rather than from chemical substances. The benefit of using antibodies, for example, is that once they are activated by a disease, they can build up resistance against it. In that sense they are used in disease prevention, not just treatment. With cheap gene sequencing, demand for drugs that stave off hereditary disease could really take off, especially as governments seek to vaccinate children to that end. Oncology vaccines are already generating huge interest, with cervical cancer recently added to many immunisation programmes. AstraZeneca has predicted that biologics will grow on average by 11% a year up to 2012, at which point they will account for 44% of the world's top 100 treatments.
The key to this research is machinery, says Tom Bulford in Red Hot Penny Shares. "Gone are the days when teams of white-coated researchers stood at laboratory tables, squinting at cells through the lens of a microscope." Today, they use machines that can identify cells at high resolution, count them at high speed and analyse their characteristics in seconds. Decoding the genomes at $1,000 may be a while off, but the companies aiding this colossal data crunch have a very strong market to attend to in the meantime. We look below at the best play in the sector.
The monstrous potential of stem cells
No one can doubt the staggering potential of stem cell research. In the last month alone, German scientists took a big step towards producing replacement body tissue using stem cells. And scientists in Newcastle have used them to develop sperm in a test tube threatening to make the male of the species redundant.
The trouble is that it's a long way from experiment to commercial treatment. And while the regulatory authorities have given a green light to the first-ever human trials using embryonic stem cells this year, we can't suddenly expect a string of medical miracles. You can blame our biology for that.
The human body consists of more than 200 mature cell types, each with its own specific function from brain to blood to muscle. Stem cells are immature cells with the chameleon-like ability to take on the characteristics of any of those 200 cell types. Embryonic stem cells hold the most potential. But it's hard to predict how these cells will behave when administered to patients. They have a nasty tendency to form malignant tumours called teratomas literally 'monster tumours' which can contain random bodily structures, such as eyes and teeth.
There has been more success with adult stem cells, but their range of uses is restricted because of their maturity. The third and best source of stem cells is the umbilical cord. Here the cells are young and easy to secure. Stem cell scientists have already used them to make 20 human-organ-based tissues, including liver, blood vessels and pancreas tissue. But again, there is no guarantee how these cells will develop.
Ironing out all these 'programming' problems with stem cells will involve scientists conducting a vast range of tests before they get anywhere near a potential commercial treatment. But as you may have gathered from the above, the sheer logistics of sourcing enough cells to do so isn't easy. For example, adult stem cells typically come from bone marrow. To extract them is an invasive and painful procedure. So for now, the best way to profit from stem cell research is to buy into firms that specialise in stem cell storage we look below at a global leader in the field.
The medical-industrial complex
President Lyndon Johnson was the Silvio Berlusconi of his day: fond of grand but ill-judged displays of manhood in front of reporters. At one press conference, Johnson reassured the assembled reporters that he had made a full recovery from routine gall bladder surgery by baring his chest. "Here ya go," bellowed Johnson, lifting his shirt to reveal a horrific scar from his navel all the way up to his chest. Cartoonists had a field day depicting the scar in the shape of Vietnam.
Medical surgery has come a long way since then. If Johnson went in for gall bladder surgery today, the surgeon would make two or three small cuts of about five to 10mm through his abdomen and then pass a long, thin telescope through to view the region on a monitor. The scar would be a lot smaller, and Johnson could have been back to work within a week.
But what hasn't changed since Johnson's administration is the way the medical industry is run. Some call it the medical-industrial complex an industry that thrives on the profit-hungry collusion of companies working in pharmaceuticals, hospitals, medical supplies and insurance. This industry has little interest in cost-effective treatment, or serving a public interest. "The ugly truth about the business of medicine is that sickness is profitable," says Andy Kessler, author of The End of Medicine. "The greater the number of treatments, procedures and hospital stays, the larger the profit." The Congressional Budget Office has pointed out that $700bn of roughly $2.3trn spent on healthcare in 2008 was wasted on treatments that did not improve health outcomes.
This flood of money to create new products, that the industry can charge ever higher fees for using, is shockingly wasteful. But it can also produce impressive new medical devices alongside the less effective treatments. As Kessler puts it, "it's like throwing spaghetti at a wall some of it sticks". What's more, medical device firms benefit from lighter regulation and lower product development times than drug developers, making them less prone to big disappointments far down the development track. Spending on new health technology already makes up as much as two-thirds of the 6% annual increase in healthcare costs, which for this year is $2.5trn. So for now, investors should follow the money and invest in medical devices.
The best plays in the sector
Genetix (Aim: GTX) made its name selling the machinery used in the 1990s for sequencing the human genome. But once the job was done, sales fell sharply. So the firm changed tack. Now it's a world leader in image screening and diagnostics. It makes lab equipment that allows scientists and clinicians to screen cells in early-stage research and design of biologics, which, as we pointed out, will account for 44% of the world's top 100 treatments by 2012. Genetix is also a leading provider of technologies for genetic analysis for prenatal and postnatal screening. It sells imaging and diagnostic equipment to big pharma, biotech and more than 1,000 diagnostic labs worldwide.
Earnings per share (EPS) has been growing at 29% a year. Increasing consolidation in the pharma industry will make it hard to maintain that pace, warns MoneyWeek's Paul Hill. But the search for biological drugs will require a huge amount of data crunching and analysis; Genetix has the tools for the job. It trades of a forward p/e of 11.5.
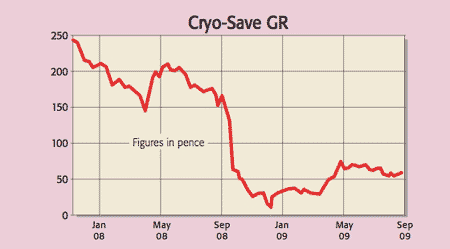
In late July, we tipped Reneuron (Aim: RENE) as a canny play on stem cells. Reneuron focuses on producing adult stem cell therapies. And two could prove themselves very quickly. Since July, Reneuron received regulatory approval to start clinical trials on its treatment for stroke patients. But its promising diabetes therapy is even closer to a breakthrough. So we still like Reneuron. But an even cannier play on stem cells is in storing them. Cryo-Save (Aim: CRYO) is the largest stem cell bank in Europe, commanding half the market. These bio-banks will be invaluable to researchers. Cryo-Save has already committed 100,000 samples to storage and last year saw a 21% jump in demand. The group turned a profit of €3.5m on turnover of €29.5m, and just reported an 11% jump in storage in the last quarter. Tom Bulford of Red Hot Penny Shares maintains a 12-month target of 140p. It trades at 58p today.
One of the most impressive medical device innovations of recent years is the Cyber Knife, from Accuray (Nasdaq: ARAY). This robotic radio-surgery gets around one of the main risks in surgical tumour removal: the risk of damaging organs due to movement during surgery. The Cyber Knife does this by moving to the rhythm of the lungs and prostate, tracking and removing tumours as the body moves. The machine has a 100% success at treating prostate cancer in studies, notes Paul Hill, and has a huge potential market.
Another medical devices group that looks set to revolutionise cancer treatment is Electro-Optical Sciences (Nasdaq: MELA). Melanoma is the deadliest skin cancer, accounting for 80% of all skin cancer deaths. There are 150,000 cases reported a year in the US alone. But doctors still must rely on their own vision to decide whether a spot or wart merits a biopsy. So Electro-Optical has developed the MelaFind, a hand-held imaging 'gun' that emits ten different light wavelengths to capture images of suspect pigmented skin lesions. The device is under expedited review by the Food and Drug Administration, but in clinical studies, MelaFind's accuracy in ruling out disease proved to be 2.5 times greater than that of dermatologists. Roth Capital Partners analyst Matt Dolan thinks sales of the device could hit $10m the first year, then climb to $40m.
This article was originally published in MoneyWeek magazine issue number 451 on 4 September, and was available exclusively to magazine subscribers. To ensure you don't miss a thing, and get instant access to all our premium content, subscribe to MoneyWeek magazine now and get your first three issues free.
Get the latest financial news, insights and expert analysis from our award-winning MoneyWeek team, to help you understand what really matters when it comes to your finances.
Eoin came to MoneyWeek in 2006 having graduated with a MLitt in economics from Trinity College, Dublin. He taught economic history for two years at Trinity, while researching a thesis on how herd behaviour destroys financial markets.
-
 UK interest rates live: rates held at 3.75%
UK interest rates live: rates held at 3.75%The Bank of England’s Monetary Policy Committee (MPC) met today to decide UK interest rates, and voted to hold rates at their current level
-
 MoneyWeek Talks: The funds to choose in 2026
MoneyWeek Talks: The funds to choose in 2026Podcast Fidelity's Tom Stevenson reveals his top three funds for 2026 for your ISA or self-invested personal pension